IP-Protected Lipids Redefining Nucleic-Acid Therapeutics
Delivery
AldexBIO’s lipids are reshaping LNPs and successful therapeutic delivery
Platform
Unique drug delivery platform that produces highly diverse novel lipids with high efficiency.
Novelty
Ability to synthesize billions of novel next-generation lipids for therapeutic delivery.
Protection
Multi layer IP protection allowing freedom to operate.
Team
EU-based team with decades of experience in chemistry and drug development.
The AldexBIO Platform
Powerful platform features
Enables fast design and synthesis of novel lipids that are customizable for specific use cases.
Novel: New-generation lipids with unique features.
Tailored for purpose: Customized lipid design for specific use cases.
High throughput lipid optimization: Rapidly design, test, and customize lipids.
Freedom to operate: Drug development with no risk of infringement on existing IP.
The AldexBIO Platform
Protected by multiple layers of IP
Six international patent applications filed from 2022 till 2024, protecting the AldexBIO lipids and the proprietary AldexBIO technology.
The AldexBIO Patent Portfolio is Well Separated from Competitor Patent Applications Published in 2019-2024
Key word search “ionizable lipids”
or “cationic lipids”
Formidable
FTO Challenge
Partially related to AldexBIO
proprietary motifs
Manageable
FTO Challenge
Within AldexBIO
chemical space
Perfect
FTO Situation
Unique Capabilities
Designed for optimization
Designed to support data-driven lipid optimization processes for therapeutic delivery.
Unique Capabilities
SAR and performance prediction
The high throughput of the AldexBIO platform allows synthesis of close structural homologues, SAR analysis and performance prediction.
Unique Capabilities
Targeting
Lipids from the AldexBIO platform deliver excellent extra-hepatic targeting results with outstanding target to liver ratios.
Unique Capabilities
In vitro transfection
Lipids from the AldexBIO platform show excellent in vitro transfection results in therapeutically relevant cells.
Toxicology and Safety
Lipids delivered by the AldexBIO platform are tested in various toxicity relevant in vitro assays and in vivo models.
AldexBIO LipexSil® Lipids Tested with Various in vitro and in vivo Toxicology Models with Promising Results
HepG2 cell viability assay
180 of 240 lipids tested
77 less toxic than Industry Standards.
Human T-cell viability assay
35 of 240 lipids tested
9 comparable to Industry Standards.
Impact on cytokine
levels in mice
5 of 240 lipids tested
no critical or “no go” observations.
In vivo transfection studies
47 lipids in 126 LNPs tested
no mortality, no loss of bodyweight,
no clinical observations.
Toxicity testing of LNPs in mice
5 of 240 lipids tested
no critical or “no go” observations.
Exploratory toxicity study in mice
3-4 lipids, 5 animals per group
In planning.
Toxicology and Safety
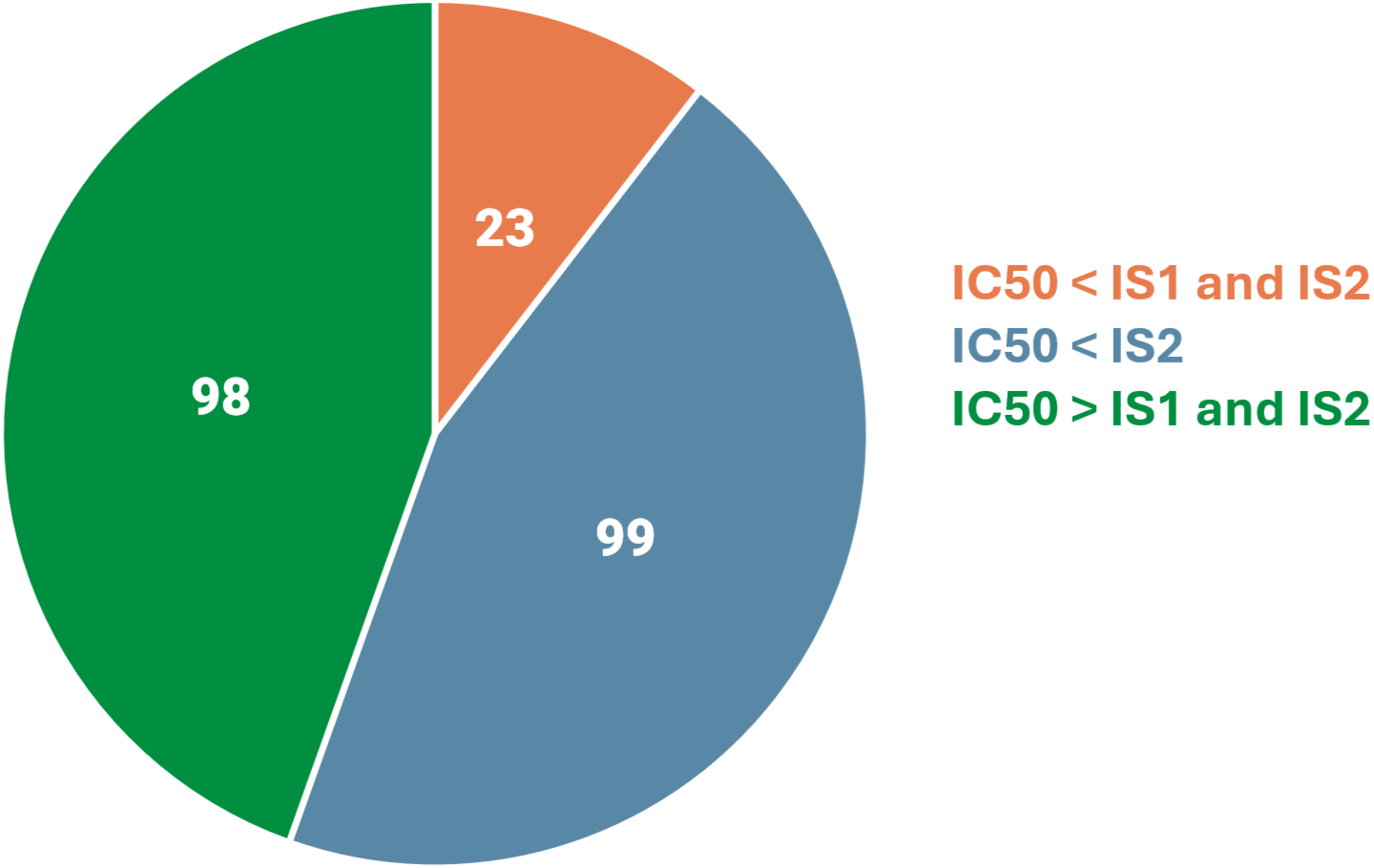
The AldexBIO platform delivered a large number of lipids that outperform Industry Standard lipids in cellular toxicity results.
220 out of 256 LipexSil Lipids Tested
Lipids with low toxicity.
Promising in vivo toxicity and tolerability.
Convincing in vitro toxicity results.
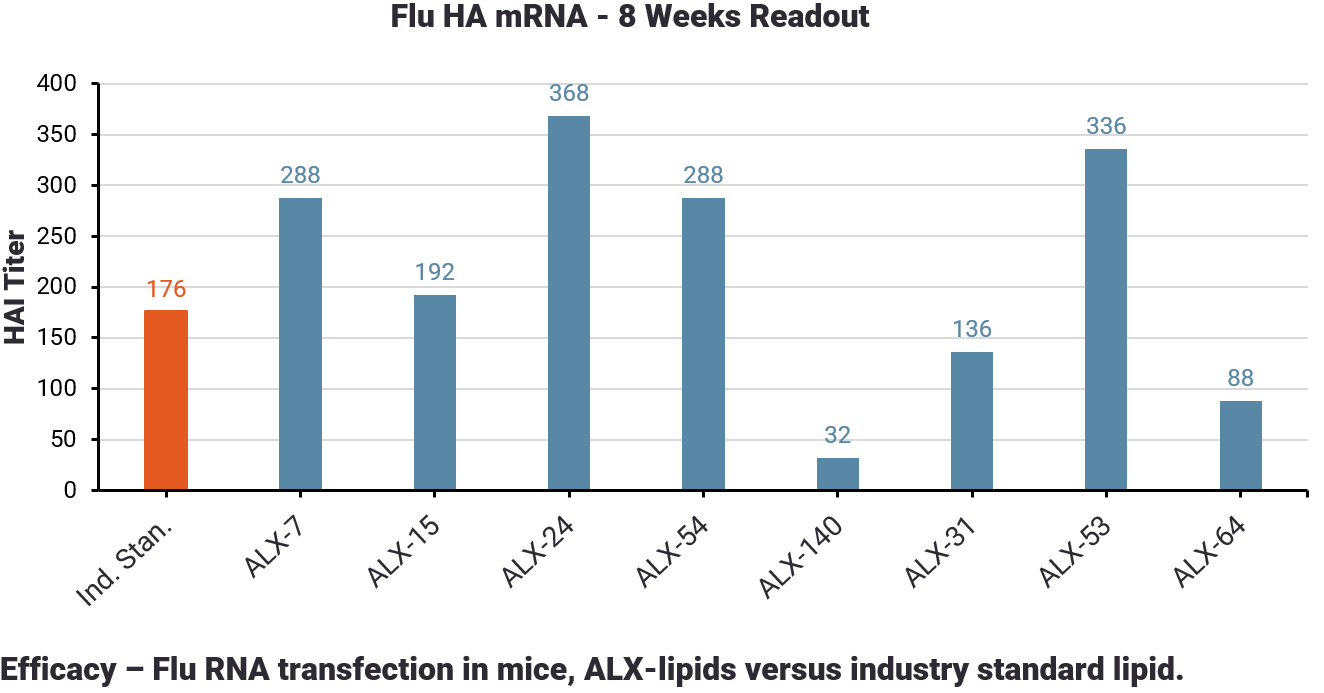
Transfection Efficacy
LNPs containing AldexBIO lipids show in vivo transfection results comparable or superior to Industry Standards.
Transfection Efficacy
LNPs containing AldexBIO lipids show in vivo transfection results comparable or superior to Industry Standard.
Transfection Efficacy
In an in vivo immunization model AldexBio lipids demonstrated superiority compared to an industry standard.
The AldexBIO Platform
Novel lipids with unique properties.
Multiple layer IP protection.
Application specific design.
Iterative optimization.
Scalable technology.
Unique properties.
Low in vitro toxicity.
Well tolerated
in vivo.
Excellent in vitro transfection.
Excellent in vivo transfection.